INTRODUCTION
The red bar.
It is the universal symbol of 21st-century anxiety. When that tiny sliver of light on your smartphone turns from white to crimson, your behavior changes. You dim your screen, you close your background apps, and you begin a desperate, subconscious hunt for a wall outlet. We are a civilization tethered to the grid by a thin chemical thread, yet almost none of us understand the invisible miracle happening inside that flat, silver pouch in our pockets.
Every time you send a text, stream a movie, or floor the accelerator of an electric vehicle, you are triggering a silent, atomic-scale migration. Inside your battery, billions of lithium ions are being shoved across a chemical frontier, forced to move back and forth in a process scientists call “The Rocking Chair” effect.
But why lithium? Why not lead, or iron, or gold? The answer lies in the volatile, energetic nature of the third element on the periodic table, a metal so reactive it can burst into flame upon touching water, yet so efficient that it has enabled the mobile revolution, the drone age, and the death of the internal combustion engine.
We take for granted that energy can be “bottled.” We assume that electricity, a force that naturally wants to strike from the sky in a bolt of lightning, can somehow be tamed, folded into a thin sheet, and tucked into a pocket next to our thigh.
This isn’t just about gadgetry; it’s about a masterpiece of chemical engineering that earned its creators the Nobel Prize. It’s a story of microscopic “apartments” where atoms live, liquid bridges they must cross, and the inevitable decay that eventually turns every high-tech device into a paperweight.
In this deep dive, we are going to shrink down to the molecular level to witness the frantic journey of the lithium ion. We will explore the delicate balance of the anode and cathode, the secret role of the electrolyte, and the future tech that promises to make “low battery anxiety” a relic of the past.
Grab your charger, this is how we bottled lightning.
TABLE OF CONTENTS
- The “Rocking Chair” Analogy: Battery Basics
- The Cast of Characters: Anode, Cathode, and Beyond
- The Atomic Dance: A Step-by-Step Breakdown
- Real-World Examples: From Smartphones to Teslas
- The Advanced Layer: The Chemistry of Intercalation
- Why Batteries Die: The Tragedy of Degradation
- Common Myths About Lithium-Ion Batteries
- The Future: Solid State and Beyond
- Interesting Facts Section
- FAQ Section
- Conclusion
The “Rocking Chair” Analogy: Battery Basics
Before we talk about chemicals and voltages, let’s use an analogy. Imagine a house with two rooms: a Living Room and a Bedroom. Between them is a hallway with a very specific kind of door.
- Charging: When you plug your phone into the wall, you are effectively acting as a “mover.” You are forcing all the “people” (Lithium Ions) out of the Living Room, down the hallway, and into the Bedroom. They don’t really want to be in the Bedroom, it’s cramped and they’re packed in tight but you’ve used the electricity from the wall to force them there.
- Discharging: Once you unplug your phone and start using it, the “people” realize the door is open. They hate the crowded Bedroom. They naturally want to rush back to the Living Room where they have more space and can relax.
As they rush back through the hallway to the Living Room, they have to turn a “turnstile.” This turnstile captures their movement and converts it into the electricity that powers your screen, your CPU, and your speakers.
In the battery world, this back-and-forth movement is why lithium-ion batteries are called “Rocking Chair Batteries.” The energy isn’t “created”, it is stored by pushing ions to one side and released as they swing back to the other.
2. The Cast of Characters: Anode, Cathode, and Beyond
To understand the system, you need to know the players. A lithium-ion battery consists of four primary components, each with a vital, life-or-death job.
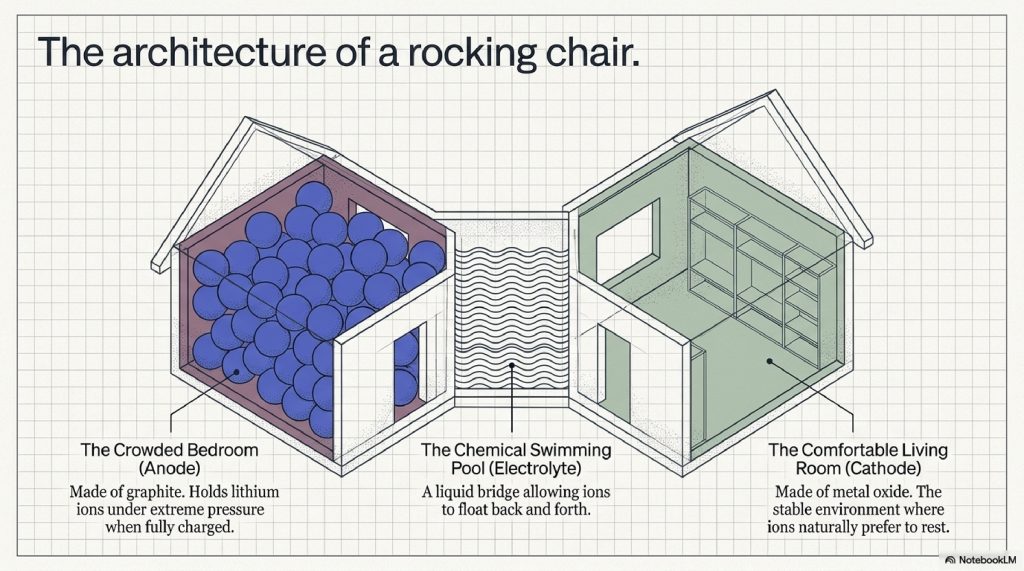
The Anode (The Crowded Bedroom)
The anode is the negative electrode. In most modern batteries, it’s made of graphite, the same stuff in your pencil. On a microscopic level, graphite looks like a stack of egg cartons. Its job is to hold the lithium ions while the battery is fully charged.
The Cathode (The Comfortable Living Room)
The cathode is the positive electrode. It is usually made of a metal oxide (like Lithium Cobalt Oxide, $LiCoO_2$). This is where the ions want to go to rest. The cathode’s chemical structure determines how much energy the battery can hold and how much power it can put out.
The Electrolyte (The Hallway)
Lithium ions can’t just fly through the air. They need a medium to move through. The electrolyte is a liquid or gel filled with lithium salts. Think of it as a “chemical swimming pool” that allows ions to float from one side to the other.
The Separator (The Security Guard)
This is the unsung hero. The separator is a thin, porous plastic film that sits between the anode and cathode. It allows the tiny lithium ions to pass through but blocks the much larger electrons.
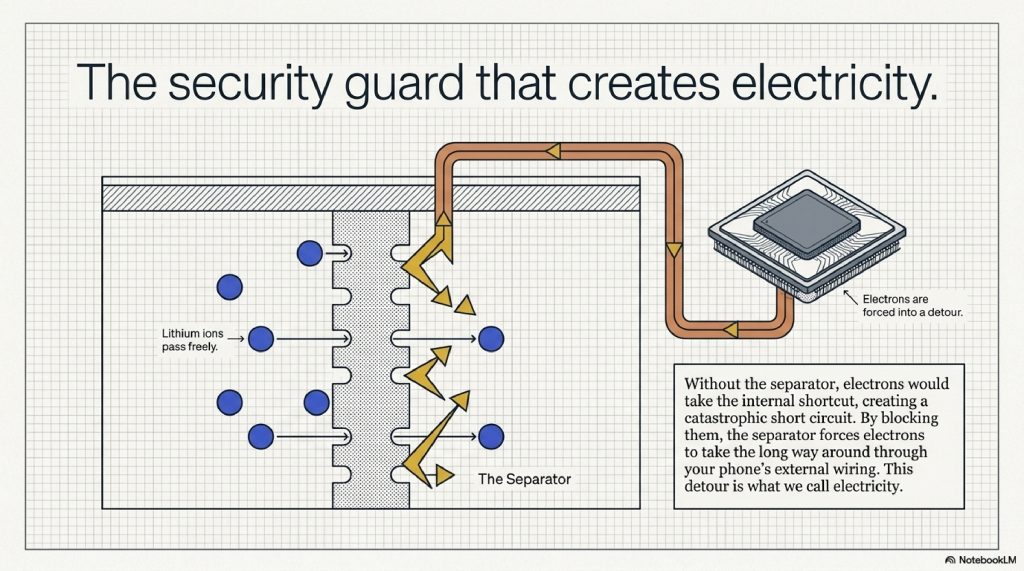
Wait… what?
If the electrons could just walk through the separator, they would take the shortcut, create a massive spark, and the battery would explode (a short circuit). By blocking the electrons, the separator forces them to take the “long way around” through your phone’s wiring. This detour is what we call electricity.
3. The Atomic Dance: A Step-by-Step Breakdown
Let’s follow a single lithium ion through a full cycle.
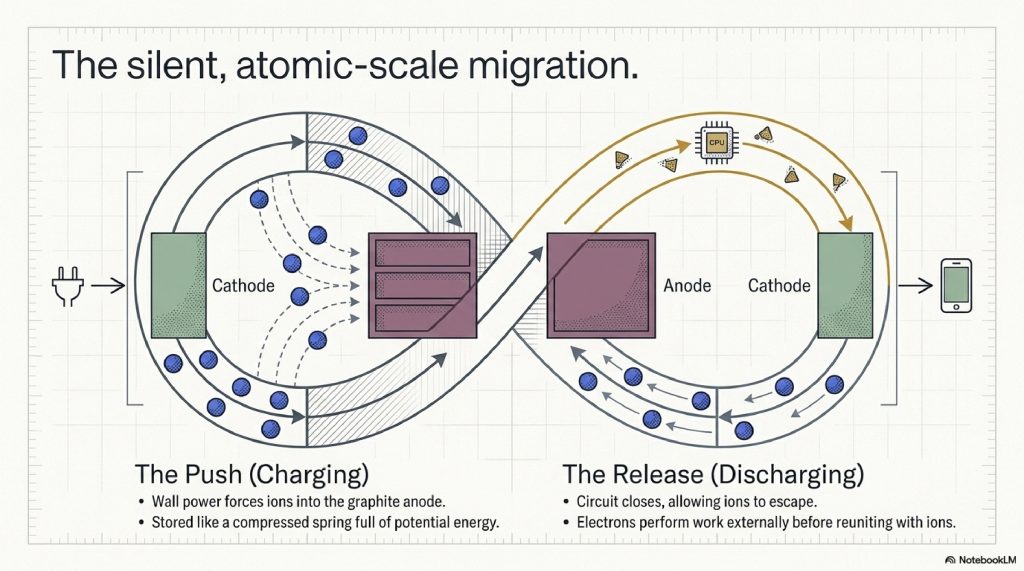
Step 1: The Charge (The Push)
You plug in your device. The charger provides an excess of electrons to the Anode. This creates a powerful negative charge that attracts the lithium ions. The ions leave their comfortable “apartments” in the Cathode, swim through the Electrolyte, squeeze through the Separator, and wedge themselves into the graphite layers of the Anode.
Step 2: The Stasis (Full Battery)
The battery is at 100%. The ions are packed into the graphite anode like a compressed spring. They are full of “potential energy.” They are unstable and desperate to leave.
Step 3: The Release (Discharge)
You open TikTok. The circuit closes. The lithium ions finally get their chance to escape. They rush back through the electrolyte toward the cathode.
Step 4: The Work (Powering the Device)
As the ions move internally, the electrons which are blocked by the separator are forced to flow through the external circuit of your phone. They pass through the processor, doing work (calculating pixels), before meeting up with the lithium ions at the cathode.
4. Real-World Examples: From Smartphones to Teslas
While the chemistry is similar, the “packaging” changes the game.
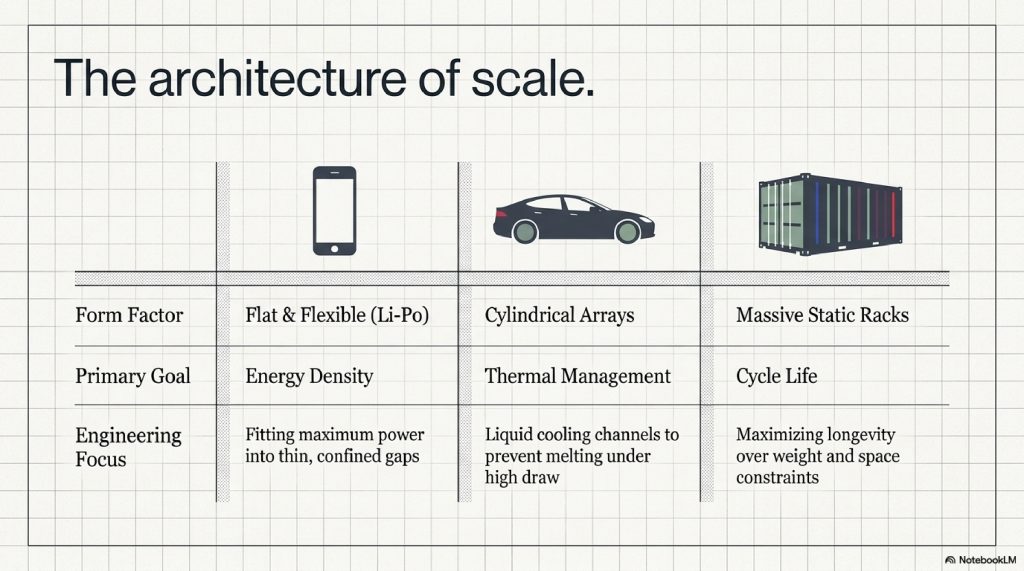
- The Smartphone: Uses a Lithium-Polymer (Li-Po) design. These are flat and flexible, designed to fit into the thin gaps behind your screen. They prioritize “energy density” fitting as much juice as possible into a tiny, light footprint.
- The Tesla / EV: An electric car doesn’t have one big battery; it has thousands of small “cylindrical cells” (looking like overgrown AA batteries) linked together in a massive pack. These prioritize “thermal management.” Because they pull so much power, they need liquid cooling channels to prevent them from melting.
- The Power Grid: Massive shipping containers full of lithium batteries are now being used to store solar energy during the day so it can be used at night. Here, weight doesn’t matter, but “cycle life” (how many years it lasts) is everything.
5. The Advanced Layer: The Chemistry of Intercalation
For the intelligent reader, the secret “sauce” of the lithium-ion battery is a process called Intercalation.
In older batteries (like the lead-acid one in your car), the electrodes actually dissolve and reform during the chemical reaction. This is messy and wears out quickly.
In lithium-ion batteries, the lithium ions do not react with the electrodes. Instead, they intercalate meaning they slide into the microscopic gaps between the layers of the host material.
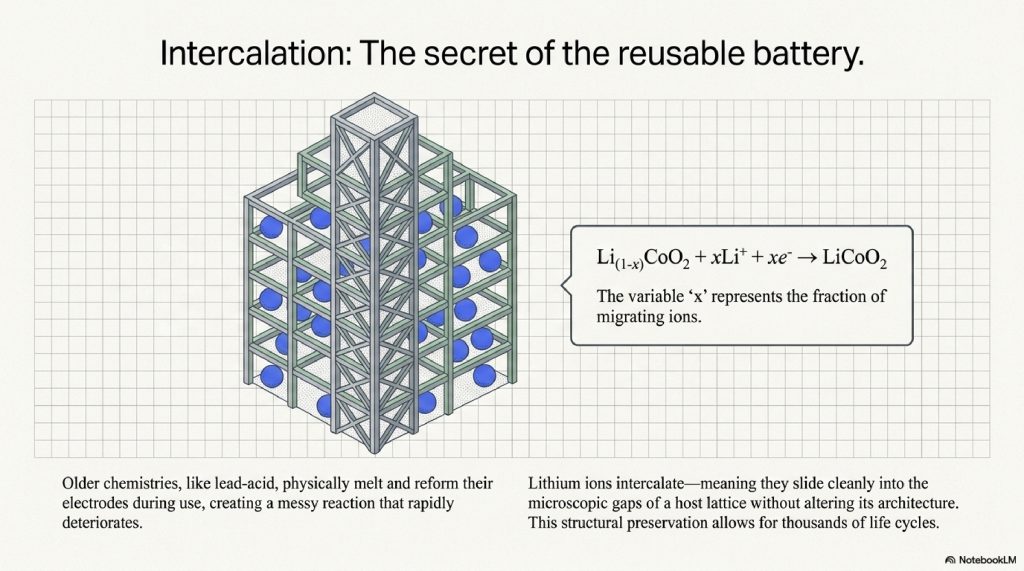
The Crystalline Lattice
Imagine the cathode as a skyscraper made of cobalt and oxygen atoms. The lithium ions are like tenants that move into the empty floors. When they move in, they don’t change the structure of the building; they just fill the empty space.
This is represented by the formula for the discharge reaction:
The “x” represents the fraction of lithium ions moving. Because the physical structure remains (mostly) unchanged, we can do this thousands of times. This is why lithium-ion batteries can be recharged, whereas an alkaline AA battery is a one-way trip.
6. Why Batteries Die: The Tragedy of Degradation
Nothing lasts forever, and in the world of batteries, heat is the enemy of time. There are three main reasons your battery life drops after a year:
- The SEI Layer Build-up: Every time you charge, a thin film called the Solid Electrolyte Interphase (SEI) forms on the anode. Think of it like “battery cholesterol.” Over time, this film gets thicker, making it harder for ions to move through.
- Lattice Stress: Even though intercalation is “clean,” the physical act of ions pushing into the graphite causes the material to expand and contract. Over hundreds of cycles, the “micro-apartments” begin to crack and crumble.
- Dendrites: This is the scary one. If you charge too fast or in the cold, lithium atoms can clump together on the surface of the anode instead of sliding inside. They grow into tiny, microscopic spikes called dendrites. If a dendrite grows long enough to pierce the separator and touch the cathode… boom. The battery enters “thermal runaway.”

7. Common Myths About Lithium-Ion Batteries
Myth 1: You should let your phone die to 0% before charging.
- The Truth: This was true for old Nickel-Cadmium batteries (the “memory effect”). For Lithium-Ion, this is actually harmful. Deep discharges put more stress on the battery. It’s better to keep it between 20% and 80%.
Myth 2: Charging overnight will “overcharge” and explode your phone.
- The Truth: Modern smartphones have “Charge Controller” chips. Once the battery hits 100%, the phone effectively “disconnects” the battery and runs directly off the wall power.
Myth 3: Putting a battery in the freezer recharges it.
- The Truth: Cold actually increases internal resistance. While heat kills batteries, extreme cold makes them temporarily unable to provide power. Keep your batteries at room temperature for the best life.

8. The Future: Solid State and Beyond
The lithium-ion battery is approaching its physical limit. To get more range out of EVs and more days out of phones, we need a paradigm shift.
Solid-State Batteries
The holy grail. Instead of a liquid electrolyte (which is flammable), we use a solid piece of ceramic or glass.
- The Benefit: They can’t catch fire, they charge in minutes, and they hold 2-3x more energy.
- The Challenge: They are currently incredibly expensive to manufacture at scale.
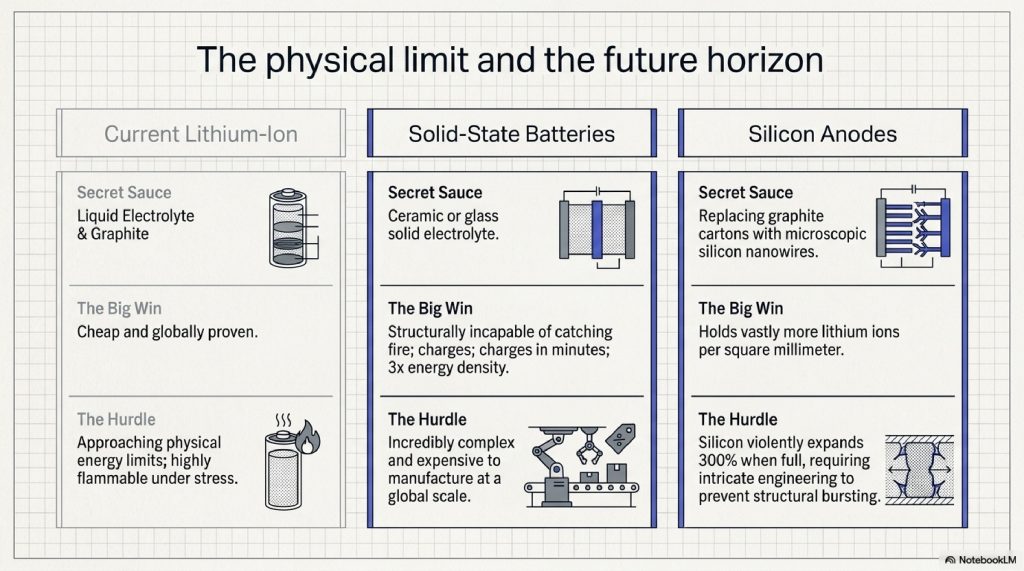
Silicon Anodes
Remember the graphite “egg cartons”? If we replace graphite with silicon, we can hold much more lithium. The problem? Silicon expands by 300% when it’s “full,” which causes the battery to literally explode like a balloon. Scientists are working on “nanowires” to solve this.
9. Interesting Facts Section
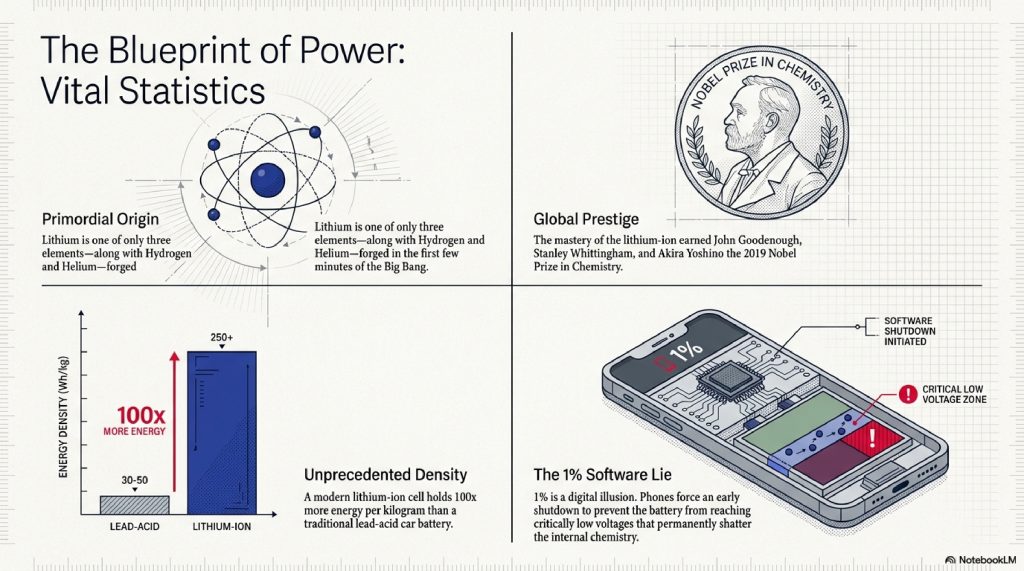
- The Nobel Prize: The 2019 Nobel Prize in Chemistry was awarded to John Goodenough, Stanley Whittingham, and Akira Yoshino specifically for the development of lithium-ion batteries.
- Lithium’s Origin: Lithium is one of the only elements (along with Hydrogen and Helium) created in the first few minutes of the Big Bang.
- Energy Density: A lithium-ion battery holds about 100 times more energy per kilogram than the lead-acid battery in your car.
- The 1% Lie: Your phone often lies to you. When it says “1%,” there is actually more energy left, but the phone shuts down to prevent the battery from reaching a “critically low” voltage that could permanently damage the chemistry.
10. FAQ SECTION
Q: Why do lithium batteries swell up like pillows?
A: This is usually due to “outgassing.” If the electrolyte decomposes due to heat or age, it turns into gas. The sealed pouch traps the gas to prevent a fire, causing it to bloat. Do not poke it.
Q: Can I use a high-wattage charger on a device that came with a low-wattage one?
A: Yes. The device “pulls” power; the charger doesn’t “push” it. Your phone’s internal controller will only take the maximum amount of power it can safely handle.
Q: Why does my phone get hot while charging?
A: Internal resistance. Moving ions through a liquid isn’t 100% efficient. Some of that kinetic energy is lost as heat. Fast chargers generate more heat because they are forcing ions through faster.
Q: Is it safe to use a phone with a cracked screen?
A: The screen is fine, but if the crack extends to the battery casing, moisture can enter. Lithium reacts violently with moisture in the air.
Q: How many cycles does a typical battery last?
A: Most consumer lithium-ion batteries are rated for 300 to 500 “full” cycles before they drop to 80% of their original capacity.
Q: What is the “best” way to store a battery I’m not using?
A: Store it at 50% charge in a cool, dry place. Storing it at 100% or 0% for long periods causes the fastest chemical breakdown.
Q: Why are they so hard to recycle?
A: They are “chemical sandwiches” glued together. Separating the lithium, cobalt, and copper requires intense heat or strong acids, which is often more expensive than just mining new materials.
Q: Why did Samsung Galaxy Note 7s explode?
A: It was a manufacturing defect where the separator was too thin. When the battery expanded slightly during use, the electrodes touched, causing a short circuit.
Q: Does fast charging damage the battery?
A: A little bit. The heat generated by fast charging accelerates the growth of the SEI layer and dendrites, but for most people, the convenience outweighs the minor loss in total lifespan.
Q: Can lithium batteries be shipped on planes?
A: Only in carry-on luggage. If a battery catches fire in the cabin, flight attendants can put it out. If it catches fire in the cargo hold, the automatic fire suppression systems often cannot stop a lithium fire.
11. INTERNAL LINKING SUGGESTIONS
- How Does SpaceX Reuse Rockets? The Science of Falcon 9
- How Banks Catch Fraudulent Transactions in Seconds
- How GPS Works: The Science of Satellites and Location
12. CONCLUSION
The lithium-ion battery is the silent engine of the modern age. It is a masterpiece of balance, a device that holds a volatile, flammable metal in a state of perpetual, controlled tension.
We often think of progress in terms of software, pixels, and AI, but none of that exists without the ability to carry energy with us. The transition from the steam engine to the internal combustion engine changed where we could go; the transition to the lithium-ion battery changed who we are. It made us mobile. It made the world’s information available at the touch of a thumb, regardless of whether we were standing in a skyscraper or a desert.

As we look toward a future of electric flight and renewable grids, the humble lithium ion remains the protagonist of our story. It is a reminder that even our most “magical” technology is ultimately rooted in the simple, elegant dance of atoms.
The next time you see that 1% icon, don’t just reach for your charger. Take a second to appreciate the billions of ions that just spent the last twelve hours rushing through a microscopic hallway to keep your world bright. They’ve earned their rest.
Comment